 The detection limit of a Western blot is determined by a number of factors, but it is important to make sure that there is enough protein for detection in the sample prior to loading. Is there sufficient antigen present in the sample for detection ? Bovine IgG may interact with the antibody due to homologous epitopes of the related species. 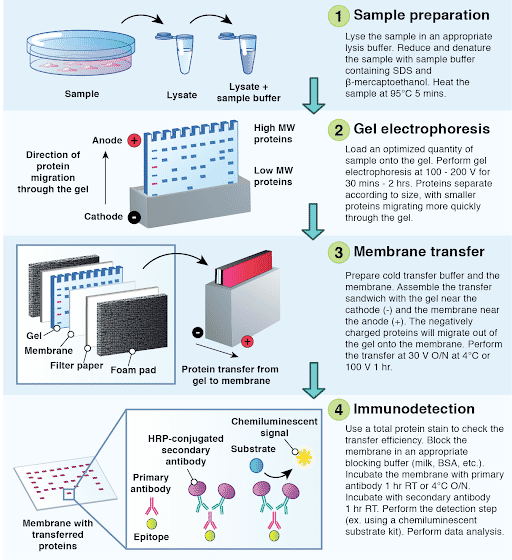
Especially when using anti-goat or -sheep secondaries, avoid using milk or BSA in the diluent buffer. PBS/Tween 20 (0.05%) or TBS/Tween, without carrier proteins, is recommended as the secondary antibody diluent. Is the secondary antibody diluent appropriate? Increase concentration of primary and/or secondary antibody, or incubate overnight at +4 o C. Is the concentration of the antibody too low? for detecting a rabbit primary use anti-rabbit secondary. Use a secondary antibody which is specific for the species the primary was raised in, i.e. If possible run a positive control for the protein of interest to confirm the specificity of primary antibodyĪre the primary and secondary antibody compatible? No specific protein bands visible.ĭoes the primary antibody recognize the protein of interest?Ĭheck with the manufacturer of the primary antibody that it has been validated for detection of protein or epitope tag of interest by Western blot. Make sure to expel air from between the gel and membrane before transfer. The Ponceau stain will identify bubbles which have interfered with protein transfer, and appear as blank circles among the transferred proteins. Small proteins can sometimes be captured by the addition of a second membrane when blotting.Īre air bubbles trapped between the gel and the membrane when blotting? If it is suspected that large proteins have not successfully transferred from gel to membrane, Coomassie stain may be used to detect proteins remaining in the acrylamide gel. Optimization of blotting conditions may be required, reducing or increasing the duration of transfer depending on the size of proteins to be blotted. Small proteins may pass through the membrane or large proteins may fail to transfer from gel to membrane. Although Ponceau cannot be used to identify a specific protein of interest, the presence of many faint pink/red bands on the blotting membrane confirms that proteins have been separated through the gel and have transferred onto the membrane. To confirm the transfer of proteins from the gel onto the blotting membrane, Ponceau S reversible stain can be a used before the blocking step. If not, then the transfer of the proteins from the electrophoresis gel to blotting membrane may have been unsuccessful. The prestained protein marker or ladder should be visible on the membrane after transfer. 
No bands are visible on the blotting membraneĬan the protein marker be seen on the membrane? If the Western blot is not behaving as expected, our troubleshooting guide may help isolate the problem. The robust nature of the antigen-antibody interaction allows the presence of specific proteins and peptides to be detected from complex mixtures. So I don't understand how the protein could either come out the other side, or not go in at all, but the ladder be fine.Download PDF Western blotting is a staple technique of the molecular biology lab. Would one of the two observations listed above suggest something that would allow the pre-stained ladder to transfer successfully, but not the protein? The ladder transfered evenly from 250 all the way down to 15 kD. So there is no protein, but the curious thing is the pre-stained ladder came out fine. pvdf thread in this forum that not soaking in water long enough can prevent protein transfer in PVDF)Ģ) His transfer chamber, when put at the same voltage I normally run (30v) went up to something like 260 mA which is about 4 times higher than the normal mA I see with my mini-transfer chamber. I told him to pre-wet in methanol, which we did, but instead of soaking in water for 5 minutes as per the instructions, he just rinsed and put in the transfer sandwich. When he was putting together the transfer, he dunked the pvdf directly in water and acted surprised when it didnt hydrate. Two suspicious things:ġ) This professor always used nitrocellulose and we use PVDF (hybond-P). 
In fact, except for the sample preparation in sample buffer, he pretty much did everything. I ran the big gel with professor who does them a lot. However, I wanted to get more resolution so for this particular western I had run a "big" gel. In fact, I had run these exact samples already twice and it worked fine. I run mini-gels all the time and never have problems. I encountered something strange the other day with my western blot:Īfter transfer, the pre-stained ladder (bio-rad) showed up fine, but there was no protein whatsoever on the blot stained with Ponceau. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
